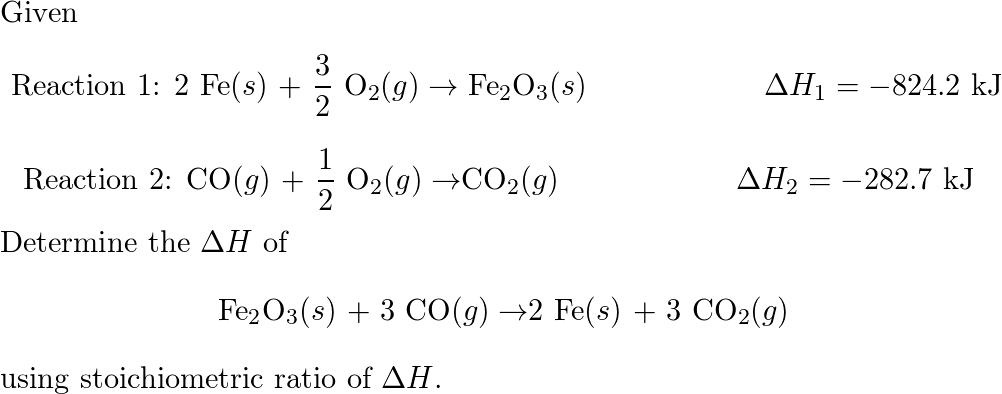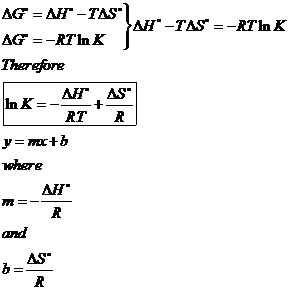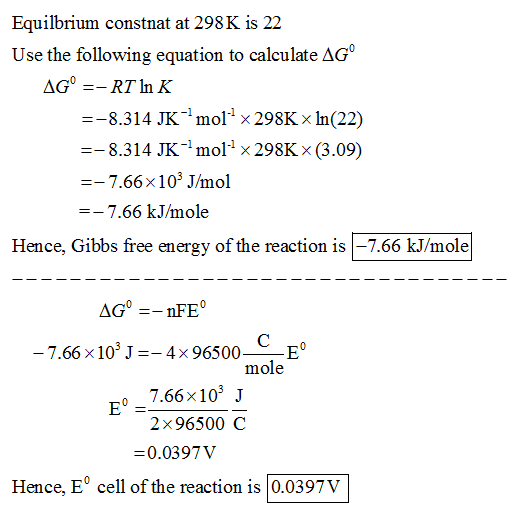✓ Solved: Calculate ΔG for the following reactions two different ways: (1) use Hess's law and the standard...

Calculate Delta G (in joule) for the reaction 2A(g) to B(g)+C(g) when mixture contains 1 mole of A, 2 moles of B and 1 mole of C at total pressure of 10
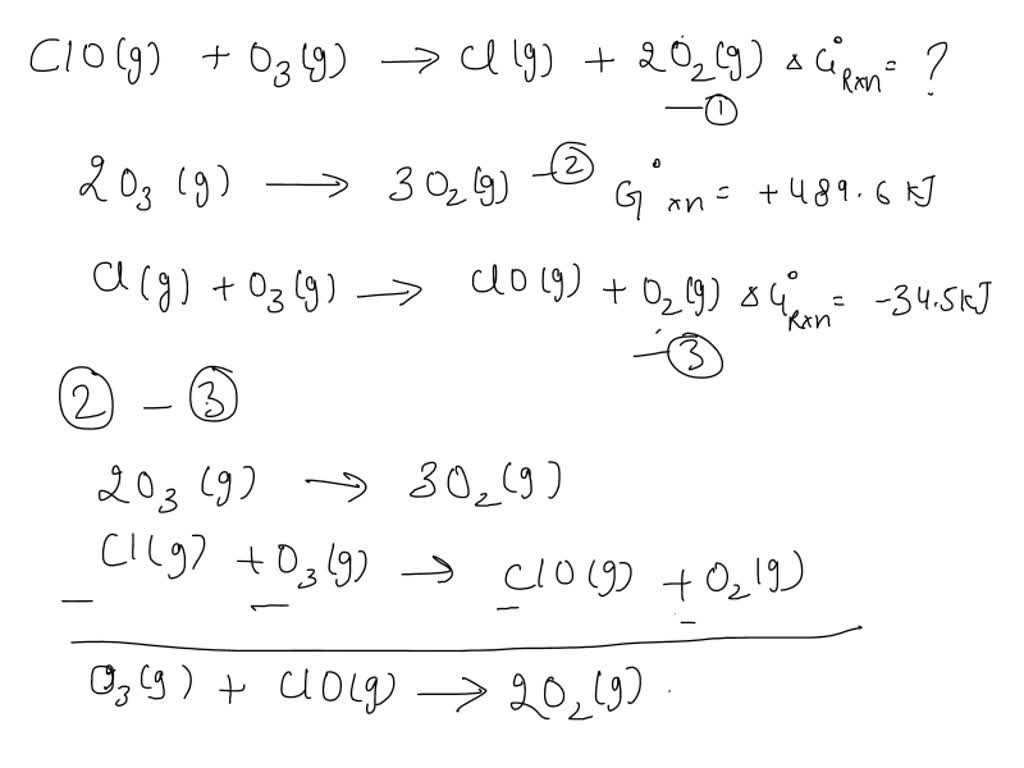
SOLVED: Use Hess's law to calculate ΔG° rxn using the following information. ClO( g) + O 3( g) → Cl( g) + 2 O 2( g) ΔG° rxn = ? 2 O

OneClass: Calculate delat G for the following reaction at 25 C . Please show steps. Calculate Delta G...

Calculate Delta G (in joule) for the reaction 2A(g) to B(g)+C(g) when mixture contains 1 mole of A, 2 moles of B and 1 mole of C at total pressure of 10