Acid and Base Production from 1.25 M NaCl, 0.2 M KCl, 0.008 M K 3 PO 4... | Download Scientific Diagram

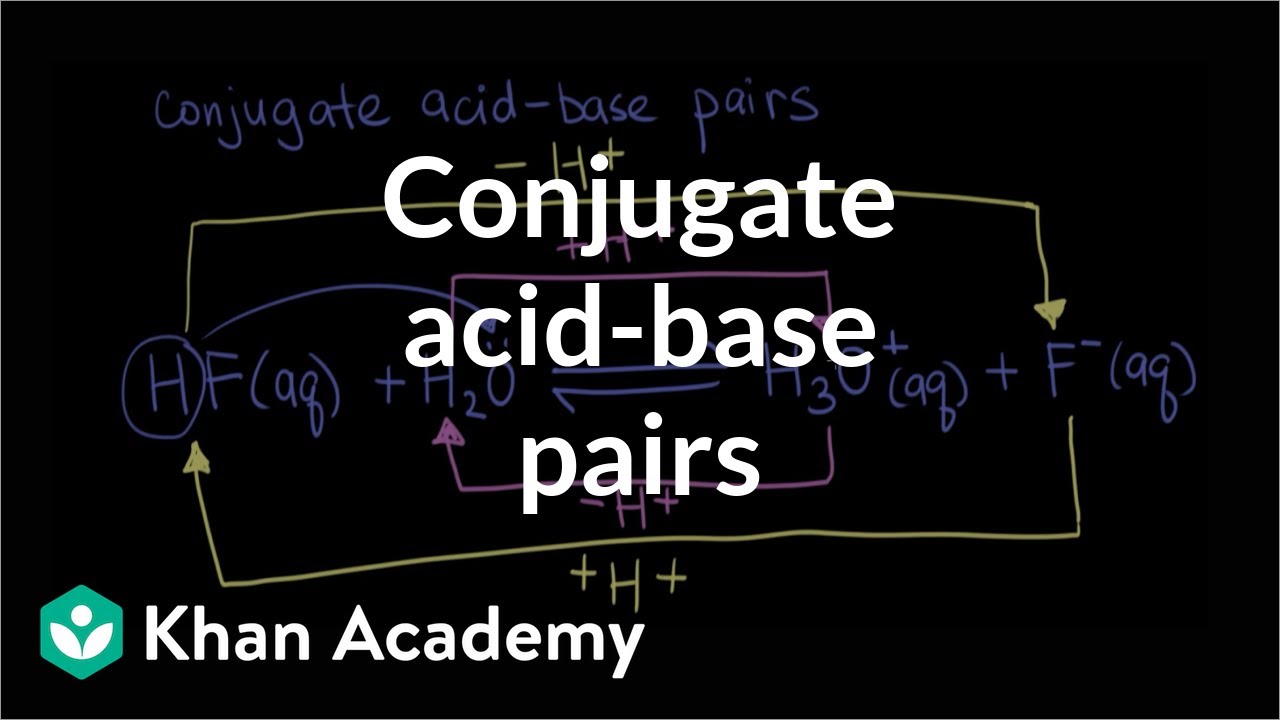
Acid-Base Properties of Salts. These salts simply dissociate in water: KCl(s) K + (aq) + Cl - (aq) - ppt download
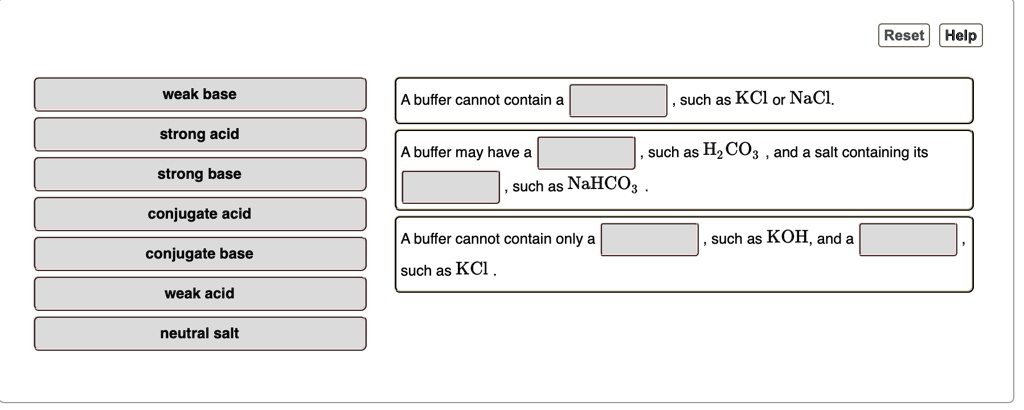
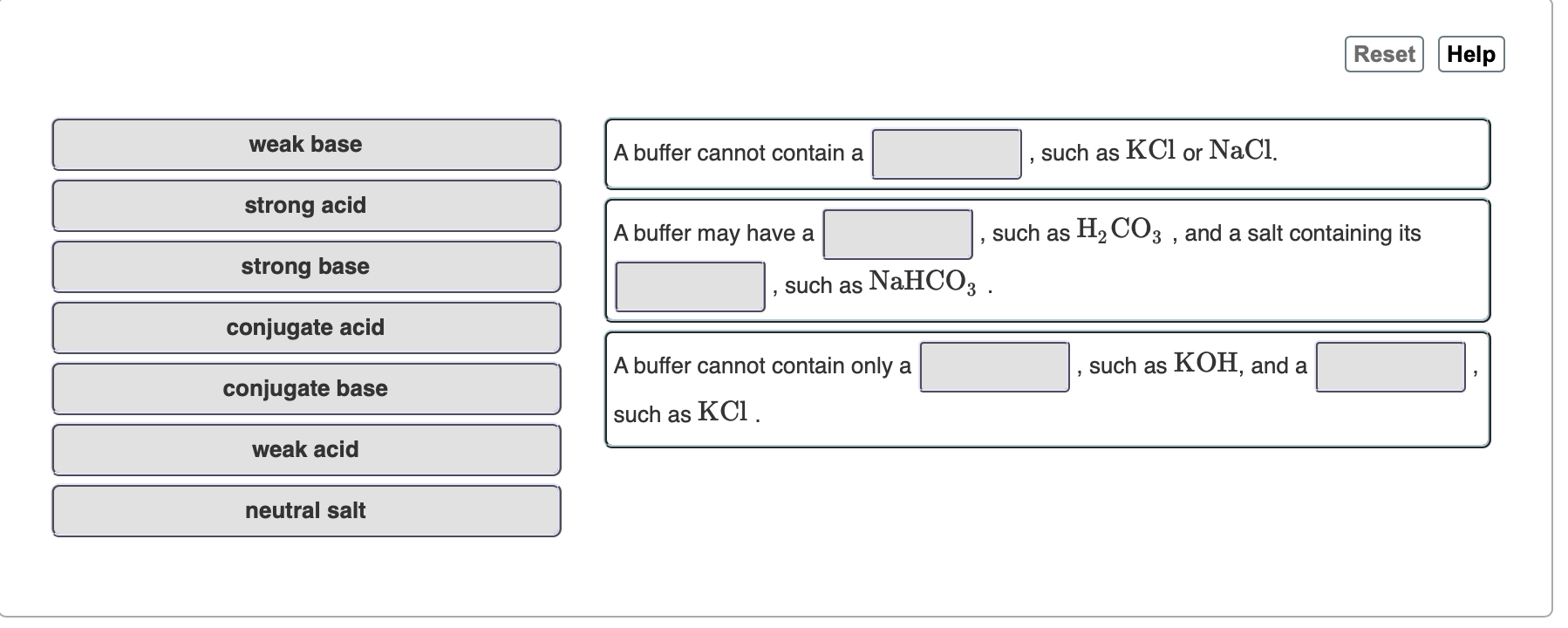
SOLVED: Reset Help weak base buffer cannot contain such as KCl or NaCl strong acid buffer may have such as HzCO: and salt containing its such as NaHCO: strong base conjugate acid

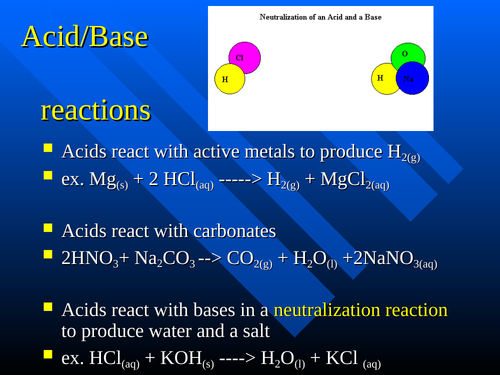
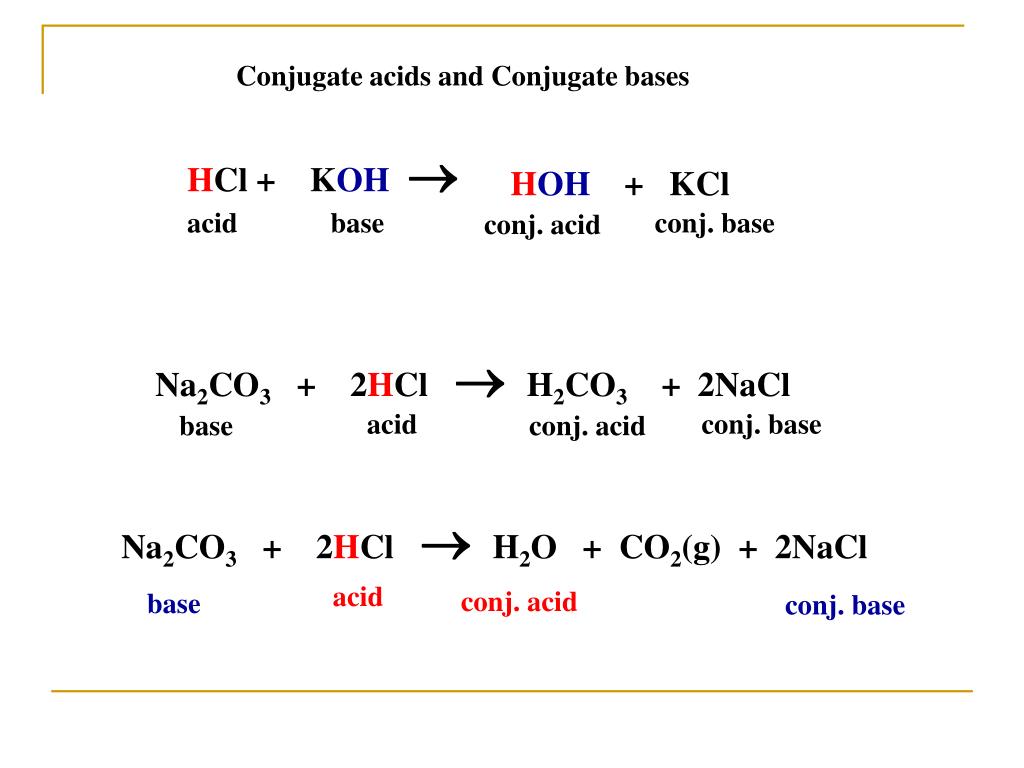
O8-#51-52 BASE ACID SALT WATER 51) KOH + HCl KCl + H 2 O COMBINE THE METAL K FROM THE BASE WITH THE NON METAL Cl FROM THE ACID TO
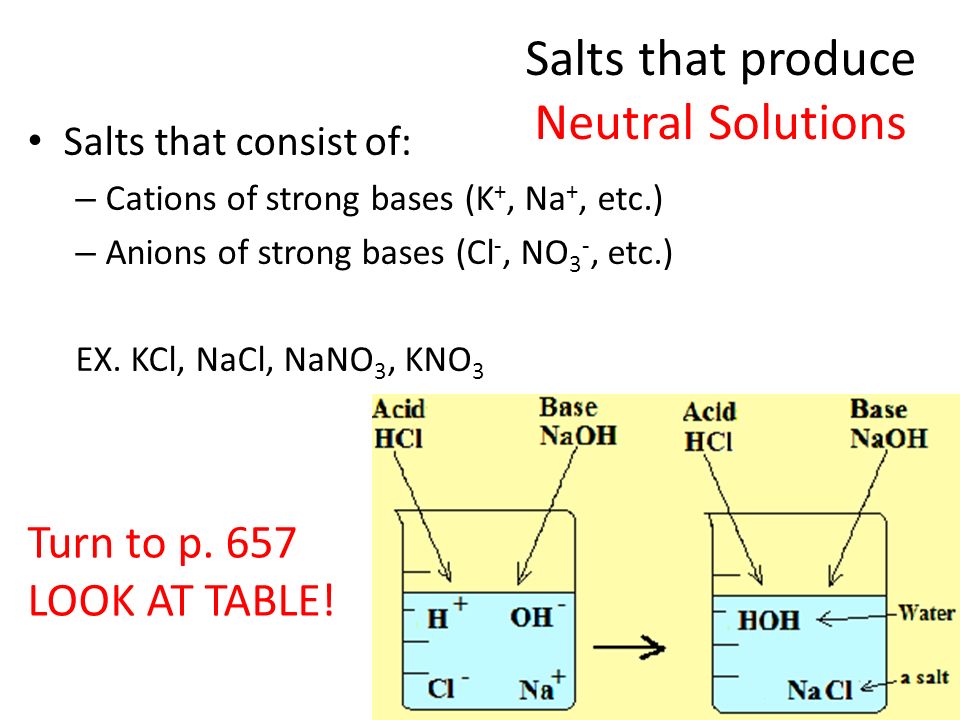
Salts as Acids and Bases Cations Sloshing Anions Sloshing Salt Ionic Compound Dissolves Ions Slosh Ions as Acids Ions as Bases Assuming that only one ion. - ppt download

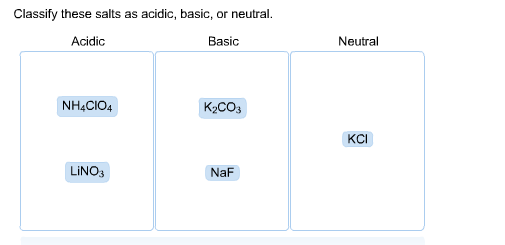
Classify these compounds as acid, base, salt, or other. NaOH, KCl, NH3, HNO3, HCOOH, CO2, NaBr, and CH3CH3? - Home Work Help - Learn CBSE Forum
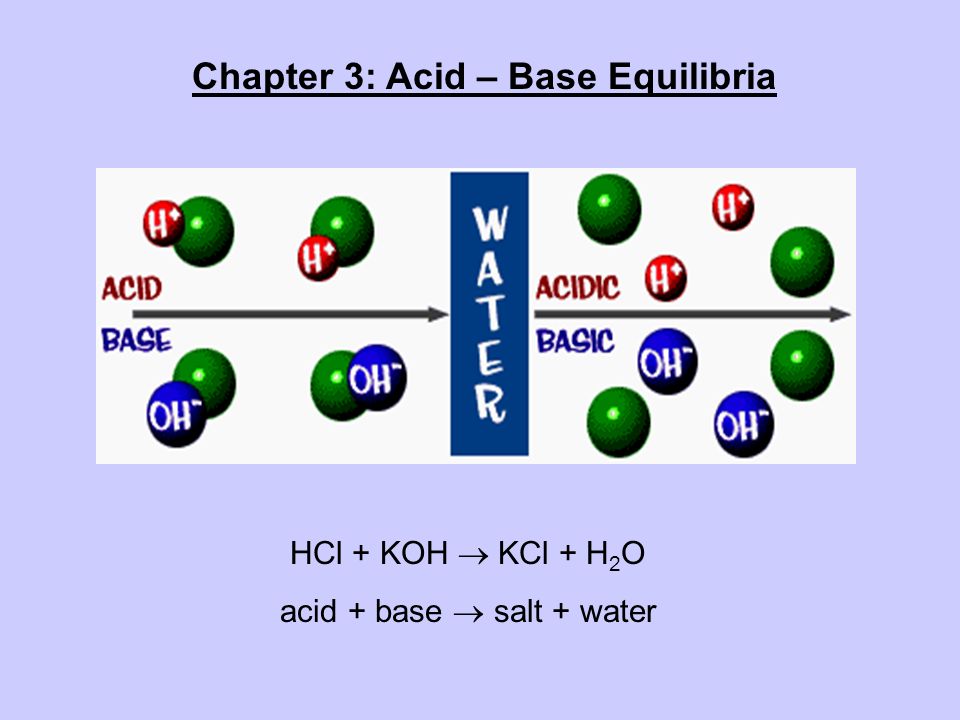
Chapter 3: Acid – Base Equilibria HCl + KOH KCl + H 2 O acid + base salt + water. - ppt download