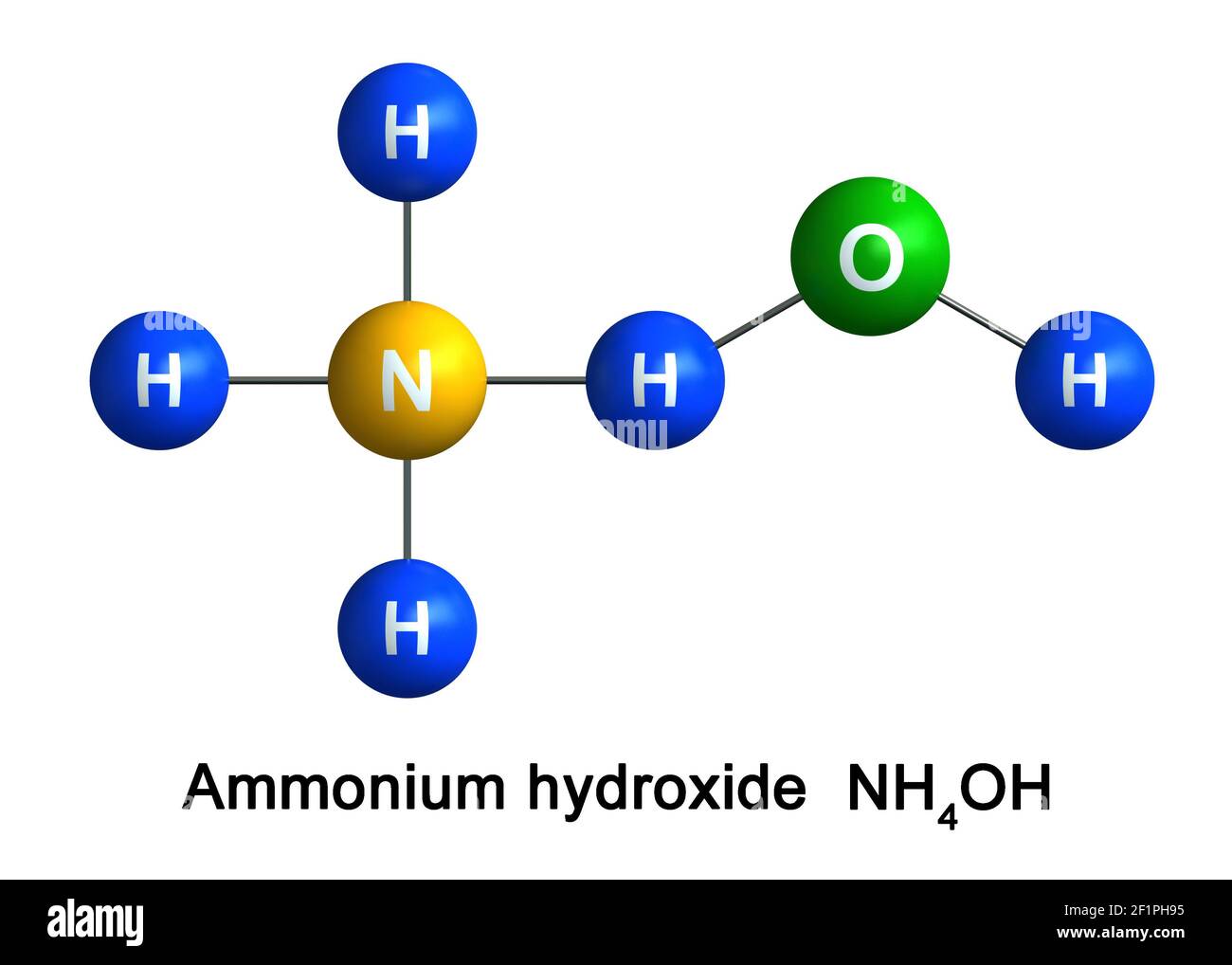
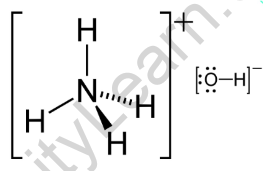
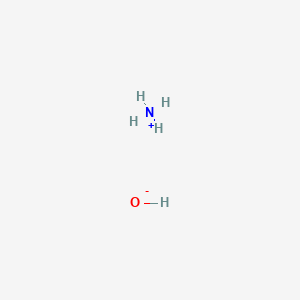
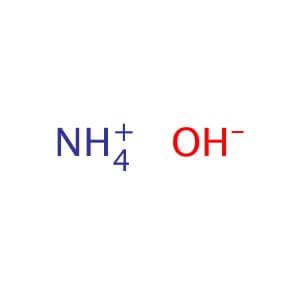
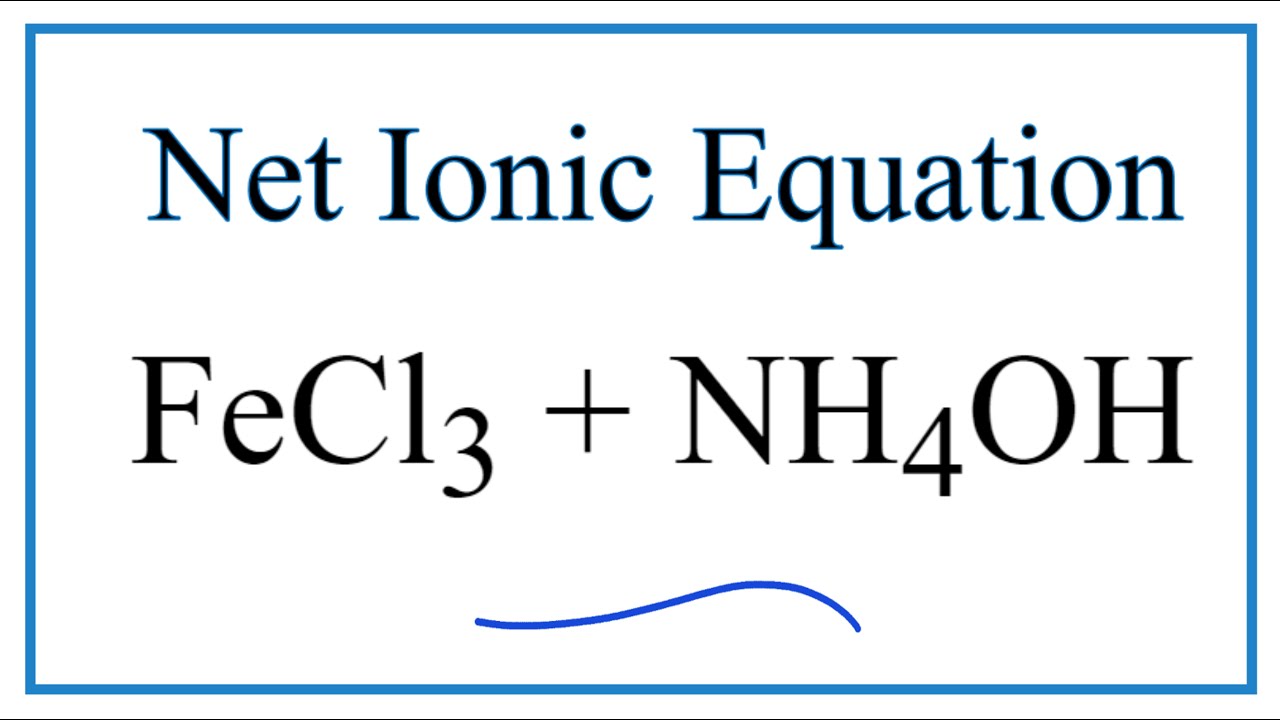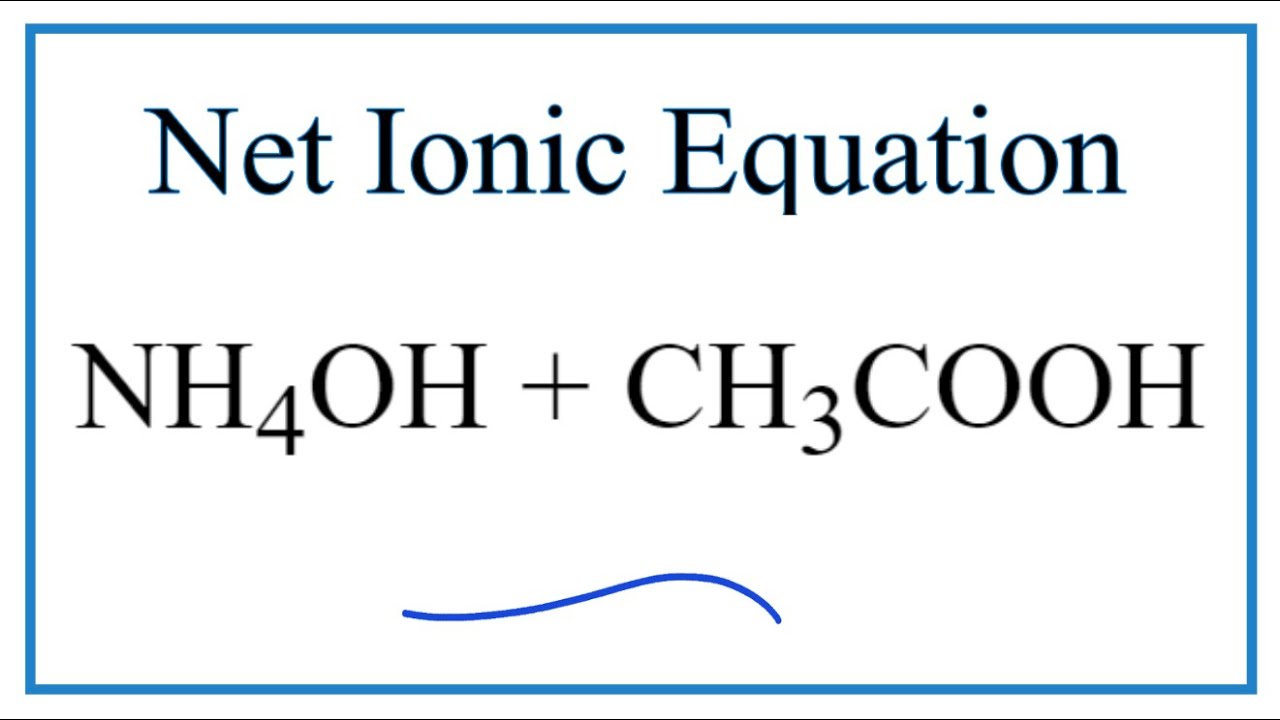The pKb value of ammonium hydroxide is 4.75 An aqueous solution of ammonium hydroxide is titrated with HCl.The pH of the solution at the point where half of the ammonium hydroxide has

Reaction of 1 with NH4OH in DMSO. (a) UV-vis spectral observations in... | Download Scientific Diagram

Question Video: The Net Ionic Equation for the Neutralization Reaction between Ammonium Hydroxide and Hydrochloric Acid | Nagwa
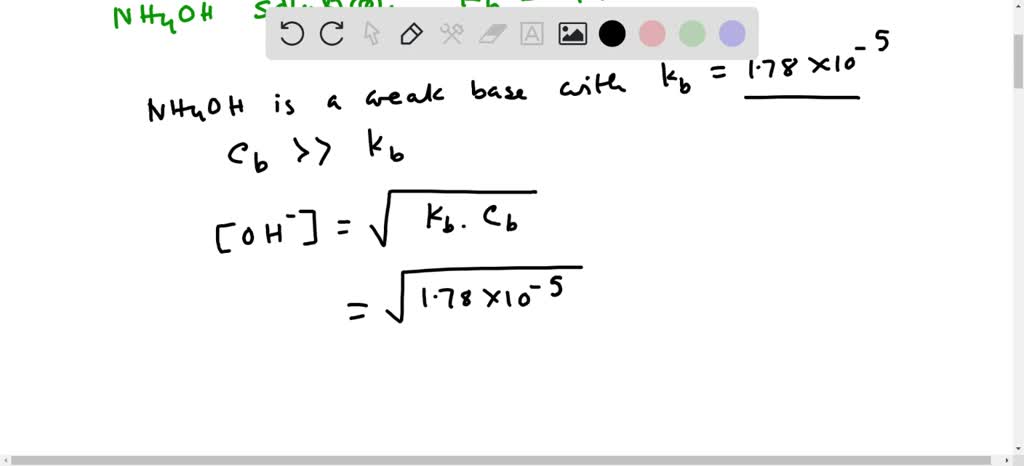
SOLVED: Answer the following equation: Ammonium Hydroxide, NH4OH, is a weak base. Calculate the pH of a 0.60 M solution of ammonium hydroxide. Kb = 1.78 x 10^-5.




![ANSWERED] When aqueous solutions of NH4OH(aq) and Cu... - Organic Chemistry ANSWERED] When aqueous solutions of NH4OH(aq) and Cu... - Organic Chemistry](https://media.kunduz.com/media/sug-question/raw/52914656-1658983365.4892888.jpeg)